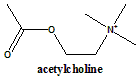
Suramin, which was originally developed for treating trypanosomiasis and onchocerciasis, inhibits the NAD +-dependent deacetylase activity of sirtuins by inducing sirtuin dimerization. AGK2, which was reported as a SIRT2 selective inhibitor, shows more than 10-fold selective inhibition relative to SIRT1 and SIRT3. remove acetyl groups to and from target lysine residues within histones, respectively, have been the most exten- sively studied at both the functional and. Functional groups are important concepts because they behave in similar ways no matter what else is present in the molecule. Kinetic analysis suggests that EX-527 binds to the nicotinamide-binding site. A high-throughput screen revealed a number of indole compounds including EX-527, which selectively inhibits SIRT1 over SIRT2. Tenovin-1 and its water-soluble analog tenovin-6 induce p53 acetylation their cellular targets were determined to be SIRT1 and SIRT2. The prefix methyl- with its locant 1 indicates that a methyl group is at. Salermide was designed based on the structure of sirtinol by molecular modeling and inhibits both SIRT1 and SIRT2 more effectively than sirtinol. This example illustrates a functional class name, and is composed of two. Acetyl chloride and acetic anhydride are commonly used. This process involves the substitution of an acetyl group for an active hydrogen atom. It is usually carried out in the presence of a base such as pyridine, dirnethylaniline, etc. Cambinol is a splitomicin-related β-naphthol that is more stable than splitomicin and increases p53 acetylation showing antitumor activity in BCL6-expressing Burkitt’s lymphoma. The introduction of an acetyl functional group into an organic compound is known as acetylation. Sirtinol and splitomicin are identified as the first small molecule sirtuin inhibitors that affect telomere silencing in yeast. Hydrogen bond interactions are drawn in dotted lines. The opposite chemical reaction is called deacetylation it is the removal of the acetyl group. ( B) A model proposed from the HDAC8 structure, in which the other histidine residue (red) plays an essential role in the electron transfer. It describes a reaction that introduces an acetyl functional group into a chemical compound. The active site zinc ion is coordinated by three residues (two aspartic acids and one histidine). Initially, two tandem histidine residues (H131 and H132) were proposed to function as Asp-His charge relay systems, typical of serine proteases such as chymotrypsin and chymotrypsinogen in the enzyme reaction. One of these catalytic histidines (red) facilitates nucleophilic attack at the substrate carbonyl by activating a water molecule coordinated with the zinc ion. The HDLP catalytic core consists of a tubular pocket, a zinc-binding site, and active-site residues (in bold) of a tyrosine (Y297) and two histidines (H131 and H132) that make hydrogen bonds to two aspartic acids (D166 and D173). ( A) A model proposed from the HDLP structure. Two models for the catalytic mechanism of the Zn-dependent HDAC reaction have been proposed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
